Briumvi (ublituximab-xiiy) MFR

On this page
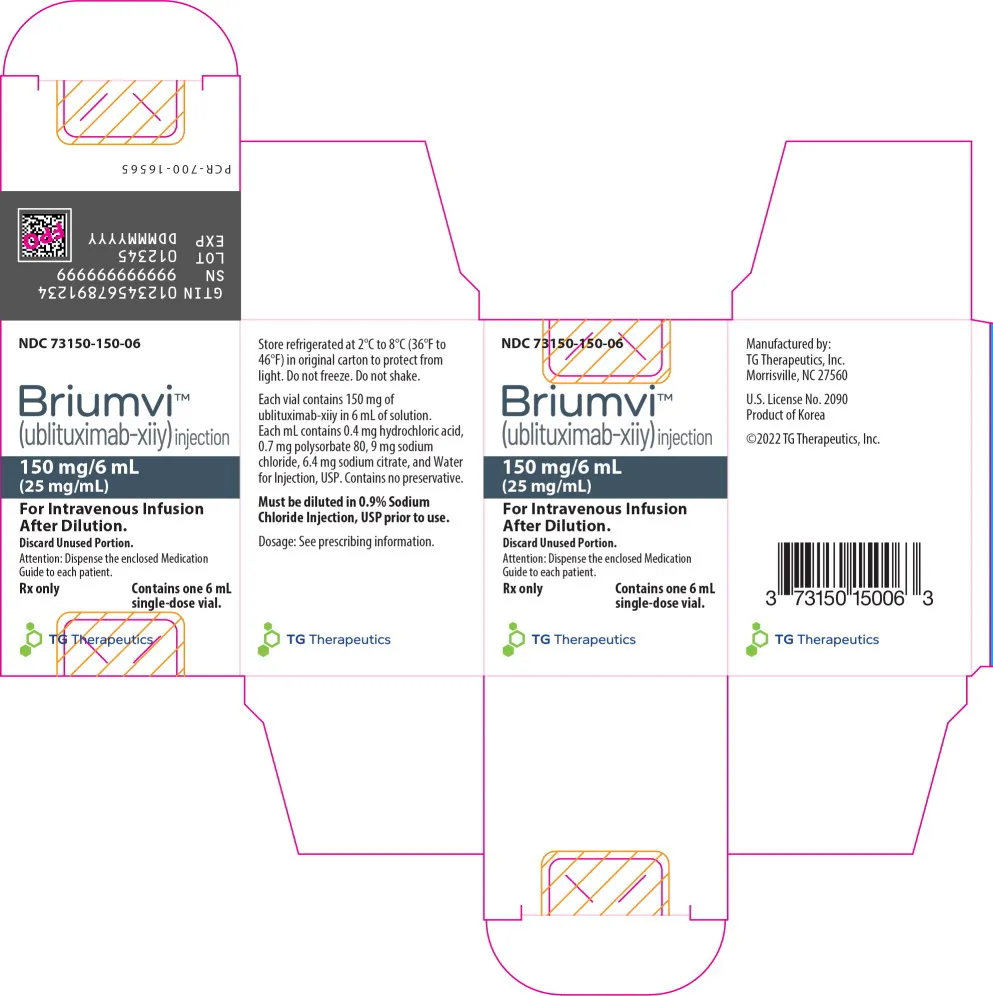
Briumvi (ublituximab-xiiy) MFR
1. Drug Identification
- Brand Name: BRIUMVI®
- Generic Name: ublituximab-xiiy
- Classification: Anti-CD20 monoclonal antibody for multiple sclerosis treatment
2. Dosage Forms and Strengths
- Dosage Form: Solution for intravenous infusion
- Strength: 150 mg/6 mL (25 mg/mL) in a single-dose vial
3. Storage Conditions (Unopened Vials)
- Store refrigerated at 2°C to 8°C (36°F to 46°F)
- Keep in original carton to protect from light
- Do not freeze
- Do not shake
4. Preparation and Administration Instructions
Pre-medication Requirements
Before each BRIUMVI infusion, administer:
- Methylprednisolone (or equivalent) 100 mg IV, approximately 30 minutes prior
- Antihistamine (diphenhydramine 50 mg or equivalent), approximately 30 minutes prior
- Antipyretic (acetaminophen 650 mg or equivalent), approximately 30 minutes prior
Initial Dose (Day 1)
- Calculate required dose: 150 mg
- Withdraw 6 mL from one BRIUMVI vial (150 mg)
- Withdraw 6 mL 0.9% Sodium Chloride Injection, USP from the 250 mL infusion bag and discard.
- Dilute in 250 mL 0.9% Sodium Chloride Injection, USP
- Gently invert to mix; do not shake
- Administer over approximately 4 hours
Second and Subsequent Doses
- Calculate required dose: 450 mg
- Withdraw 18 mL 0.9% Sodium Chloride Injection, USP from the 250 mL infusion bag and discard.
- Withdraw 18 mL BRIUMVI solution from the vials (6 mL/vial).
- Dilute in 250 mL 0.9% Sodium Chloride Injection, USP
- Gently invert to mix; do not shake
- Administer over approximately 1 hour
5. Special Compounding Considerations
- No reconstitution required (solution is ready to use)
- Handle gently - do not shake vials
- Swirl gently to mix when preparing dilution
6. Diluent Information
- Diluent: 0.9% Sodium Chloride Injection, USP only
- No other diluents have been studied
7. Diluent Amount
- 250 mL of 0.9% Sodium Chloride Injection, USP per infusion
8. Reconstituted Concentration
- Not applicable (solution is provided ready to use)
9. Compatible Solutions
- 0.9% Sodium Chloride Injection, USP only
10. IV Bag Size
- 250 mL bag specified for all doses
11. Dilution Concentration Range
- Final concentration will vary based on dose:
- Initial (150 mg): 0.6 mg/mL
- Second and subsequent doses (450 mg): 1.8 mg/mL
12. Special Equipment Needs
- Use an infusion set with an in-line, sterile, non-pyrogenic, low protein-binding filter (0.2 or 0.22 micron pore size)
13. Filter Requirements
- In-line, sterile, non-pyrogenic, low protein-binding filter (0.2 or 0.22 micron pore size) required
14. Final Appearance
- Clear to slightly opalescent, colorless to slightly yellow solution
- Free from visible particles
15. Stability of Opened/Reconstituted Vials
- Single-use vials; discard unused portion
- Do not reuse vials
16. Stability of Diluted Solution
- Prepared infusion solution should be administered immediately
- If not administered immediately, store up to 24 hours refrigerated at 2°C to 8°C (36°F to 46°F)
- Allow to reach room temperature before administration
- Can be stored at room temperature for up to 8 hours, including infusion time
17. Light Protection
- Keep vials in original carton to protect from light until time of use
- Protection from light not specifically required during administration
18. Hazard Assessment
- Not classified as hazardous
- Pregnancy Category: No human data. Based on animal data, may cause fetal harm.
- Females of reproductive potential should use effective contraception during treatment and for 6 months after the last dose
19. Oncology Use
- Not indicated for oncology use
- Indicated for relapsing forms of multiple sclerosis (MS)
Reference Information
- Package Insert Revision Date: December 2022
- Reference ID: 5101565
- FDA Approval Letter Date: 2023
https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761238s000lbl.pdf