Expanding HER2-Targeted Therapy: Trastuzumab Deruxtecan in HR⁺, HER2-Low and Ultralow Metastatic Breast Cancer
A phase 3 trial tests trastuzumab deruxtecan for HR⁺, HER2-low or ultralow metastatic breast cancer after endocrine therapy, where options are limited post-CDK 4/6 inhibitors. The study compares this targeted therapy to standard chemotherapy to improve outcomes.
Background
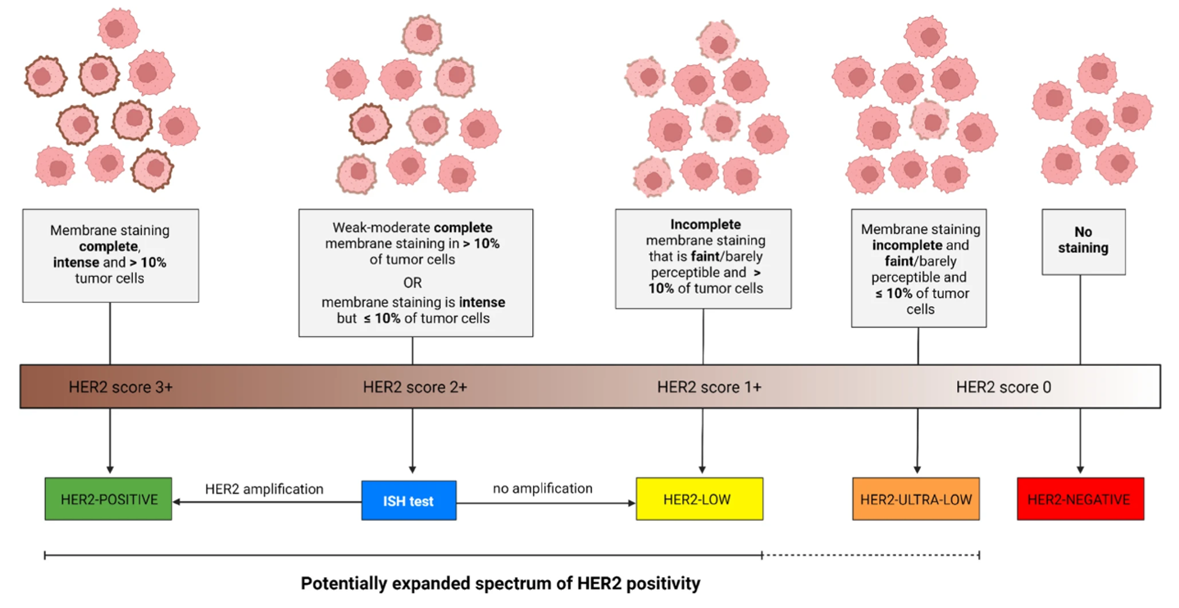
A spectrum of HER2 (human epidermal growth factor Receptor) expression exists among breast cancers that are categorized as “HER2-negative,” defined as a score on immunohistochemical (IHC) analysis of 0, 1+, or 2+ and negative results on in situ hybridization (ISH). Cancers with an IHC score of 1+ or 2+ with negative results on ISH are currently defined as “HER2-low.” More recently, it has been proposed to subdivide IHC 0 into two categories, defined according to membrane staining that is faint and is seen in 10% of tumor cells or fewer as “HER2-ultralow” or no observable staining as HER2-negative.
Current standard treatment for patients with hormone receptor positive and what is considered broadly HER2-negative metastatic breast cancer (which HER2-low and HER2-ultra low fall under) is endocrine therapy with usually a CDK (Cyclin dependent kinase) 4/6 inhibitor (ex. palbociclib, ribociclib, or abemaciclib) however the issue is that benefit of endocrine therapy–based regimens declines after exposure to CDK4/6 inhibitors due to resistance hence outcomes for these patients worsen after one or more lines of endrocrine-based therapy. Similarly with respect to progression that occurs after multiple lines of endocrine therapy or rapid progression that occurs with adjuvant or first-line endocrine therapy, conventional single-agent chemotherapy shows limited efficacy in later lines of treatment.
Trastuzumab Deruxtecan
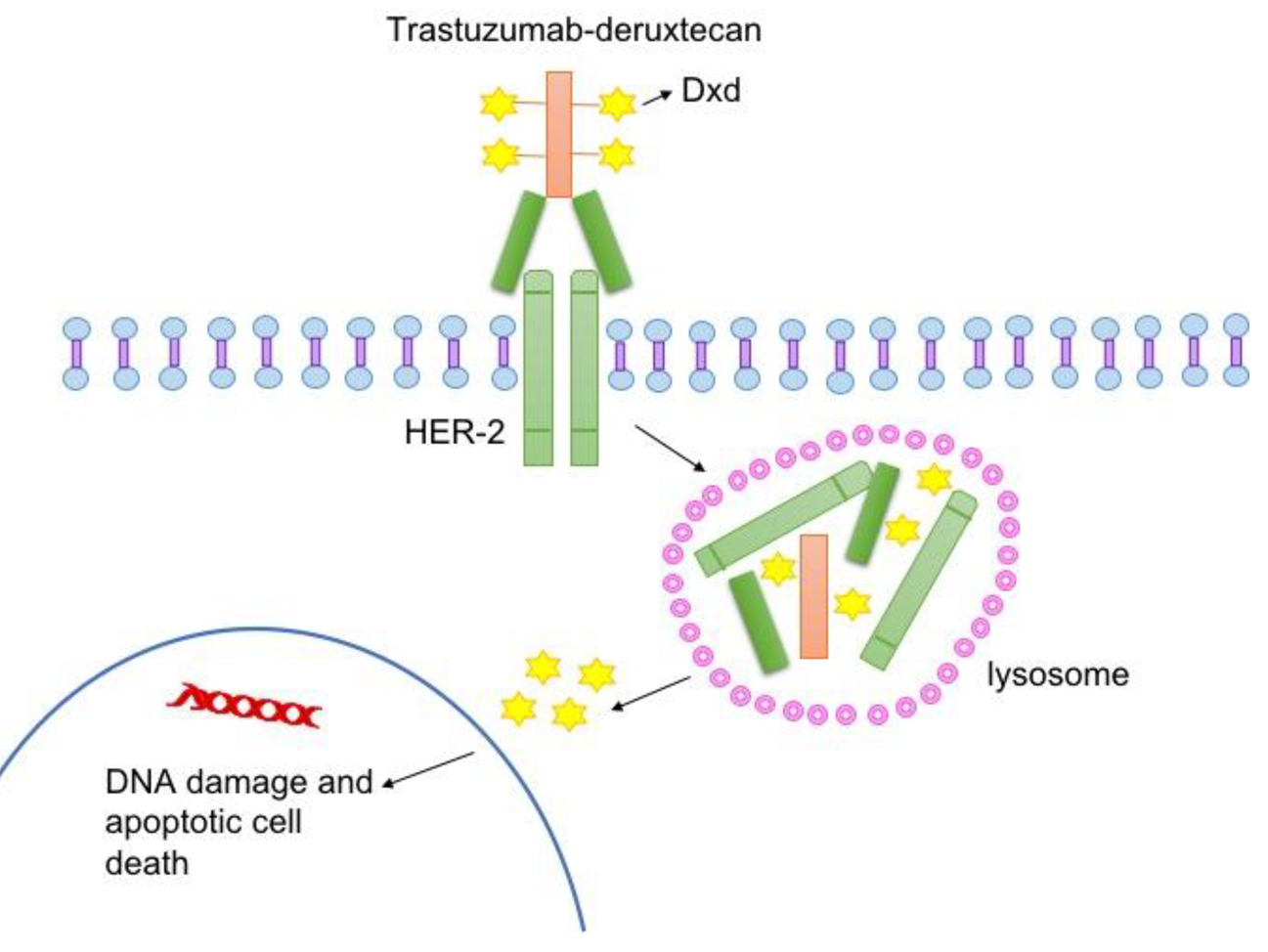
Trastuzumab deruxtecan is an antibody-drug conjugate made of humanized immunoglobulin G1 monoclonal antibody which uses the Trastuzumab mechanism to specifically target HER2 receptor on cancer cells and with deruxtecan being a tetrapeptide-based cleavable linker, once internalized by the cancer cell, cleaves lysosomal enzymes to release cytotoxic payload hence also being a potent topoisomerase I inhibitor à it interferes with DNA replication/causes double strand breaks in DNA which leads to the cancer cell’s death. In a previous DESTINY-Breast04 trial, trastuzumab deruxtecan has demonstrated efficacy in patients with metastatic breast cancer who have low expression HER2 following previous chemotherapy
Study Objective and Methods
Objective: to evaluate the efficacy and safety of trastuzumab deruxtecan as compared with the physician’s choice of chemotherapy in patients with hormone receptor–positive, HER2- low or HER2-ultralow metastatic breast cancer who had received one or more endocrine-based therapies but no previous chemotherapy for metastatic disease --> in hopes to expand the benefit from HER2-directed treatment to more patients
Trial Design
phase 3, multicenter, open-label, randomized trial
Trial Setting
324 sites in mostly Europe, Asia, and North America from August 20, 2020 to March 18, 2024
Participants
866 patients with HR+ Metastatic Breast Cancer underwent randomization and assigned 1:1 ratio to receive trastuzumab deruxtecan (5.4 mg per kilogram of body weight intravenously) once every 3 weeks or to receive the physician’s choice of single-agent chemotherapy (capecitabine, nanoparticle albumin-bound [nab]-paclitaxel, or paclitaxel) until the occurrence of disease progression
Intervention
Trastuzumab deruxtecan
Comparison
Physician's choice of chemotherapy (paclitaxel, nab-paclitaxel, or capecitabine)
Glossary
🧬 Biology & Cancer Types
| Term | Meaning |
|---|---|
| HER2 | A protein on some cancer cells that helps them grow. |
| HER2-negative | Very little or no HER2 protein on cancer cells. |
| HER2-low | Small amounts of HER2 protein (not enough for older HER2-targeted drugs). |
| HER2-ultralow | Extremely tiny amounts of HER2 or almost none. |
| Hormone Receptor Positive (HR⁺) | Cancer cells that have receptors for hormones (like estrogen) which help them grow. |
| Metastatic Breast Cancer | Breast cancer that has spread beyond the breast to other parts of the body. |
🔬 Diagnostic Tests
| Term | Meaning |
|---|---|
| IHC (Immunohistochemistry) | A lab test that uses special dyes to measure HER2 protein on cancer cells. |
| ISH (In Situ Hybridization) | A lab test that checks if cancer cells have extra HER2 genes. |
💊 Treatments
| Term | Meaning |
|---|---|
| Endocrine Therapy | Treatment that lowers or blocks hormones to slow cancer growth. |
| CDK 4/6 Inhibitors | Drugs (like palbociclib, ribociclib, abemaciclib) that block cancer cells from dividing. |
| Chemotherapy | Drugs that kill cancer cells or stop them from multiplying—can also affect healthy cells. |
| Antibody–Drug Conjugate (ADC) | A “guided missile” cancer drug that delivers chemo directly to cancer cells. |
| Trastuzumab Deruxtecan | An ADC—trastuzumab targets HER2, deruxtecan is the chemo payload. |
| Topoisomerase I Inhibitor | A chemo type that stops cancer cells from copying DNA, causing them to die. |
📊 Study Design Terms
| Term | Meaning |
|---|---|
| Phase 3 Trial | A large study comparing a new treatment with the standard approach. |
| Randomized Trial | Patients are assigned to treatments by chance to compare results fairly. |
| Open-label Trial | Both patients and doctors know which treatment is given. |
